|
9/18/2023 0 Comments 13 carbon molar mass 
Molecular Formula13CO2 Average mass45.002 Da Monoisotopic mass44.993183 Da. So how good is our atomic radius? Standard tables give the atomic radius of strontium is in the range 192-220 pm. Structure, properties, spectra, suppliers and links for: Carbon dioxide. Since 3 3=27 and 4 3 = 64, you know that the cube root of 55 will be between 3 and 4, so the cube root should be a bit less than 4 × 10 –8. Before placing in container D, check the pH with pH Universal indicator strips (Cat.No. To neutralize alkyl sulfates, add dropwise (from a dropping funnel) to concentrated ice-cool Ammonia solution (Cat.No. Alkyl sulfates are carcinogenic Take particular care to avoid inhalation and skin contact. Molecules of this compound are comprised of 13 carbon atoms, 18 hydrogen atoms, and 2 oxygen. Keep container tightly closed.ĩ Carcinogenic compounds and flammable compounds labelled "Highly toxic" or "Toxic": container F. The mass in grams of 1 mole of substance is its molar mass. 
P403 + P233: Store in a well-ventilated place. P308 + P313: IF exposed or concerned: Get medical advice/ attention. In this context, you can understand how to find molar mass and much more. In addition, the atomic mass calculator displays a pie chart for mass percentage composition by element. Remove contact lenses, if present and easy to do. An online molar mass calculator allows you to calculate the molecular weight, hill notation, nominal, and monoisotopic mass for given chemical formulas. P305 + P351 + P338: IF IN EYES: Rinse cautiously with water for several minutes. Call a POISON CENTER/doctor if you feel unwell. P304 + P340 + P312: IF INHALED: Remove person to fresh air and keep comfortable for breathing. Well to figure that out, and thats why this periodic table of elements is useful, we just have to figure out the molar mass of the constituent elements. P303 + P361 + P353: IF ON SKIN (or hair): Take off immediately all contaminated clothing. P210: Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. P201: Obtain special instructions before use. H372: Causes damage to organs through prolonged or repeated exposure. H225: Highly flammable liquid and vapour. Ph Eur and ACS.Ĭarbon disulfide for spectroscopy Uvasol®. always include the units for molecular weight (grams/mole). The most stable artificial radioisotope is 11 C, which has a half-life of 20.3402 (53) min. A few things to consider when finding the molar mass for C6H12O6 : - make sure you have the correct chemical formula. This is also the only carbon radioisotope found in nature, as trace quantities are formed cosmogenically by the reaction 14 N + n 14 C + 1 H.  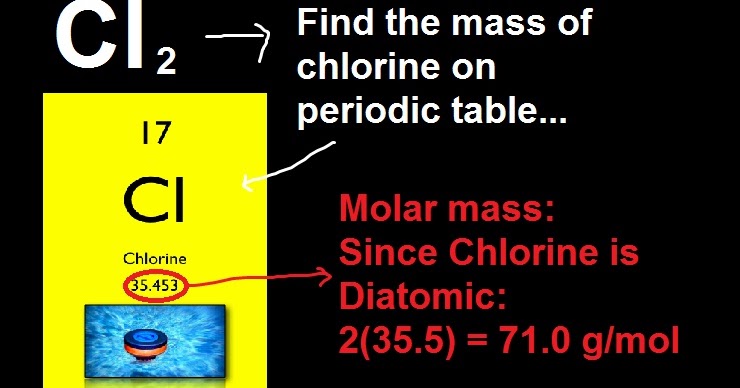
It is convenient to consider 1 mol of C 9 H 8 O 4 and use its molar mass (180.159 g/mole, determined from the chemical formula) to calculate the percentages of each of its elements: C 9 mol C × molar mass C molar mass C 9 H 8 O 4 ×. Furthermore the transmittance is specified in accordance with Reag. The longest-lived radioisotope is 14 C, with a half-life of 5.70 (3) × 103 years. To calculate the percent composition, the masses of C, H, and O in a known mass of C 9 H 8 O 4 are needed. In all specifications the minimum transmittance for 5 typical wavelengths are identified. Uvasol® solvents offer best UV transmittance. The refinement process allows a greater degree of security in applications and avoids misinterpretation of analytical results caused by traces of UV, IR and fluorescence contamination. The relative atomic mass of carbon-12 is defined as exactly 12 and the relative atomic mass of carbon-13 is 13.00335. The Uvasol® solvents range has been specially designed for spectroscopy and other applications requiring solvents of the highest spectral purity. So, it has 52.14 g of carbon, 13.13 g of hydrogen, and 34. Step 1: First, we need to convert the mass percentages into moles. 99.0 atom 13C, <3 atom 18O CAS Number: Molecular Weight: 45.00 Beilstein: 3087932 MDL number: MFCD00064513 PubChem Substance ID: 24862443. The molar mass of the chemical is also known it is 46.069 g mol 1. Accurate analytic results in UV/VIS and infrared spectroscopy depend on the use of very pure solvents for sample preparation. It is known that a chemical compound contains 52.14 carbon, 13.13 hydrogen, and 34.73 oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
